A light emitting diode (LED) is a semiconductor diode that produces light of one or more wavelengths. A semiconductor is a substance whose electrical conductivity can be changed as a result of variations in temperature, applied fields (electrical or magnetic), concentration of impurities (e.g., doping), etc. A diode is a device whereby electrical current can pass in only one direction. LEDs are an type of solid state lighting (SSL) wherein light is emitted from a solid object (e.g., a block of semiconductor or organic layers or materials) as opposed to from a vacuum or gas tube, which is the case in traditional incandescent light bulbs and fluorescent lamps. And since the vast majority of LEDs use inorganic semiconductors, the acronym LED normally represents inorganic-semiconductor-based LEDs. Some LEDs go with organic semiconductors (carbon-based small molecules or polymers), and the acronym OLEDs denotes these organic-semiconductor-based LEDs. They are very much like inorganic-semiconductor-based LEDs in that passing an electrical current through an OLED generates an excited state that can then emit light.
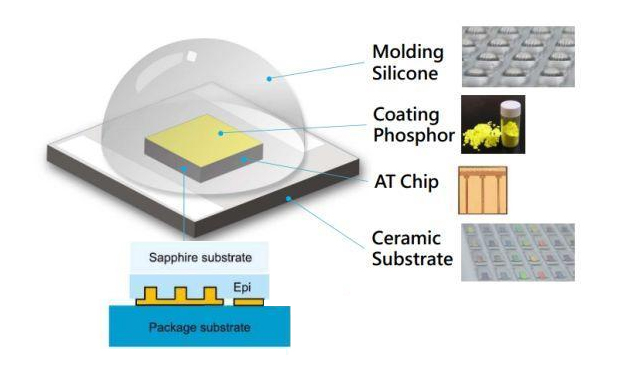
An LED incorporates semiconducting materials doped with impurities to produce a p-n junction, wherein electrical current can straightforwardly flow one directionally from the p-side (anode) to the n-side (cathode), however, not in the reverse direction. A light emitting diode emits light by exciting electrons across the band gap between a conduction band and a valence band of holes (the semiconductor active or light-emitting layer. The combo of an electron and a hole can spontaneously produce a photon at a designated wavelength or a range of wavelengths. A photon is a quantum of electromagnetic radiation or the particle which transmit light. To paraphrase, light is carried over space by photons. Color variations for visible light are generally defined by electromagnetic radiation or optical wavelengths. The wavelength of the light emitted from any particular LED is a function of the amount of energy released when an electron and a hole recombine. The color of the light (wavelength) emitted by a light emitting diode relies on the semiconductor materials of the active layers of the light emitting diode. Different wavelengths of light can be created making use of different materials by changing the bandgaps of the semiconductor layers and by forming an active layer within the p-n junction. LEDs may emit light in one wavelength range, by way of example red, amber, yellow, green, blue, etc. or may be manufactured to emit white light by employing a wide variety of different types of semiconductor materials such as, for example, III-V semiconductor materials, and II-V semiconductor materials. Single color LEDs may be fabricated applying a combination of a LED emitting light of a first wavelength and a light conversion material, for instance a phosphor, that absorbs a portion or all of the first emitted wavelength and re-emits it at a second wavelength.
Light emitting diodes can work individually or in any combinations, optionally in conjunction with one or more luminescent material (e.g., phosphors or scintillators) and/or filters, to radiate light of any preferred perceived color (including white). A phosphor is a luminescent material that produces a responsive radiation (primarily visible light) when excited by a source of exciting radiation. More often than not, the responsive radiation has a wavelength, which is generally longer, than the wavelength of the exciting radiation. Inclusion of luminescent materials in LED devices has primarily been achieved by including the luminescent materials to a clear plastic encapsulating material (e.g., epoxy-based or silicone-based material). A blue LED (typically 460 nm) surrounded by a yellow phosphor, for example cerium-doped yttrium aluminum garnet (YAG:Ce), that gives off lights in a broad band centered at 550 nm. The combination of nominally yellow light emission from the phosphor and blue light from the LED emits a light source that has a typically white appearance. Then again, an LED that emits in the ultraviolet (<400 nm) can be used to excite a blend of red, green, and blue phosphors. In a typical LED-based white light luminaire, a monochromatic LED is encapsulated by a transparent material that contains suited compensatory phosphors. The wavelength(s) of the light emitted from the compensatory phosphor is complementary to the wavelength of the light emitted by the LED to ensure that the wavelengths from the LED and the compensatory phosphor mix together to generate white light.
LED chips are typically encapsulated in a package that extracts the light and shields the chip from being damaged. The extensively recognized and commercially available "LED" that is sold (for instance) in electronics stores usually represents a "packaged" device comprised of a variety of parts. Generally speaking, an LED package consists of a substrate, an LED die (alternatively referred to as LED chip) prearranged on the substrate, and an encapsulation formed on the substrate to cover the LED die. The encapsulant encapsulates the LED chip and a portion of the carrier to protect the LED chip and expose a portion of the carrier outside the encapsulant to give a function of external electrodes. A lens is typically mounted to the LED package by using encapsulant to guide or change the nature of the light emitted by the LED. An LED die is frequently mounted on a larger substrate for heat dissipation and packaging. The substrate may contain other circuitry, such as a passive electrostatic discharge device. The package additionally has robust anode and cathode leads for being soldered to a printed circuit board (PCB).
As is well known, an LED also produces an unwanted byproduct - heat throughout the generation of light. When an electron meets a hole, it falls into a lower energy level, and produces energy in the form of light (photon, radiant energy) and heat (phonon, thermal energy). LED performance is extremely sensitive to operating temperature, which makes thermal management a vital concern in LED package and Iuminaire design. The heat is required to be systematically dissipated and/or the electrical drive power (and consequently light output) should be kept low enough, to prevent dysfunction or rapid performance degradation and/or to maintain operating efficiency. There are a selectionof techniques that have been utilized to dissipate heat from packaged electronic devices. Typical cooling systems can involve the application ofa heat sink and/or fan to cool the junction point. A heat sink is a component or assembly that delivers generated heat to a lower temperature medium. Heat sinks provide a pathway for absorbing the heat produced by the LEDs and for dissipating the heat directly or radiantly to the surrounding environment.
LEDs are low-voltage current-driven devices which implies they typically operated with direct current (DC). LEDs receive a direct current (DC) input signal or a modulated square wave input signal in order for a constant current flows through the LED chips to generate light. Therefore, integrating LEDs into residential and commercial lighting systems (which are traditionally wired to receive alternating current (AC)) necessitates that the LED be connected to a "driver" circuit which converts alternating current into a suitable direct current for the LED. The LED drivers can regulate the illumination of the LEDs by controlling the amount of current that flows through the LEDs. Aside from that, LED driving circuits can also convert power from one voltage level to another voltage level.




















































